Biomarker Services for Drug Development
Accelerate drug development with end-to-end biomarker support

How do tailored biomarker strategies accelerate project timelines?
Custom-designed assays and streamlined workflows allow your team to generate regulatory-ready data faster. By minimizing delays between discovery, preclinical, and clinical stages, Concept Life Sciences helps you make informed decisions quickly, keeping your programs on schedule.
Move from discovery to clinical validation with confidence.
Concept Life Sciences delivers tailored biomarker strategies, custom assay development, and regulatory-ready data that empower faster, smarter decisions throughout your drug development pipeline. Whether you’re identifying early disease markers or validating therapeutic response in clinical trials, our scientific experts work as an extension of your team combining innovative thinking with tailored workflows and cutting-edge technologies.
Why biomarkers matter in drug development
Biomarkers are essential to understanding how a therapeutic behaves, predicting patient response and demonstrating efficacy and safety. They support decision-making across the entire development continuum by providing evidence for:
Mechanism of action: Clarify biological pathways and target engagement.
Efficacy and safety: Evaluate therapeutic response and off-target effects.
Patient selection: Enable stratified and precision medicine approaches.
Toxicity and risk: Identify potential liabilities early.
Clinical validation: Support regulatory submissions and outcome measures.
From early discovery to clinical trial validation, biomarkers are the link between mechanism, measurement, and medicine.
Tailored biomarker solutions, built around your goals
We understand that every drug development program is different, so we co-develop biomarker strategies ensuring every result adds value to your next decision.
- Precise and scalable - supporting everything from small exploratory studies to full regulatory submissions.
- Aligned with your objectives - whether you're in discovery, preclinical, or clinical stages.
- Optimized for speed and efficiency - accelerating timelines and reducing complexity.
Download the biomarkers brochure and explore how we support your end-to-end biomarker needs.
Comprehensive biomarker capabilities
Our multidisciplinary teams have extensive experience in a comprehensive range of biomarker identification technologies and services:
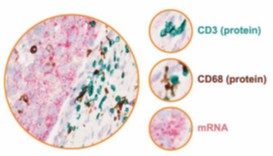
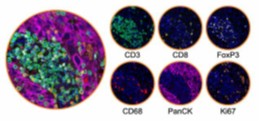
Spatial biology, histology, and image analysis: Following the guidelines set out by GCP we offer an end-to-end service enabling visualization and quantitation of protein and RNA biomarkers within a tissue context.
UCNP‑enhanced IHC (Lumito SCIZYS): An ultra-sensitive tissue imaging technology combining upconverting nanoparticle (UCNP) labeling with whole‑slide imaging to deliver background‑free, high‑contrast images across a wide dynamic range. SCIZYS enables detection of ultra‑low expressed biomarkers and separation of morphology and biomarker signal, improving objectivity and reproducibility.
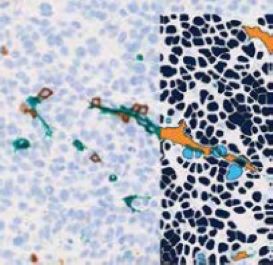
Molecular biology: Providing insight into mechanisms of action and biological pathways using an omics-based approach, provides a robust and ultrasensitive method for biomarker discovery and justification of targets for therapy
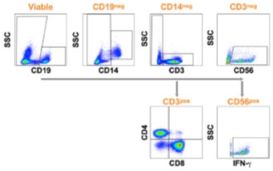
Cellular analysis: Identify phenotype specific markers and functional responses.
Secretome analysis: Multi-analyte detection to accurately identify reliable biomarkers and measure therapeutic responses. Our robust multiplex immunoassays are used for early stage-predictive toxicology, mechanism of action or assessment of therapeutic effect.
Biophysical characterization: Biophysical techniques allow the detection of biomarkers in clinical samples for early diagnosis and patient stratification. Grating-Coupled Interferometry (GCI) and Surface Plasmon Resonance (SPR) provides several advantages over conventional detection methods.
Bioanalysis: Techniques allow for targeted quantitative analysis of biomarkers with bioanalytical method validation allowing biomarker analyses to be used as end points for clinical evaluations.
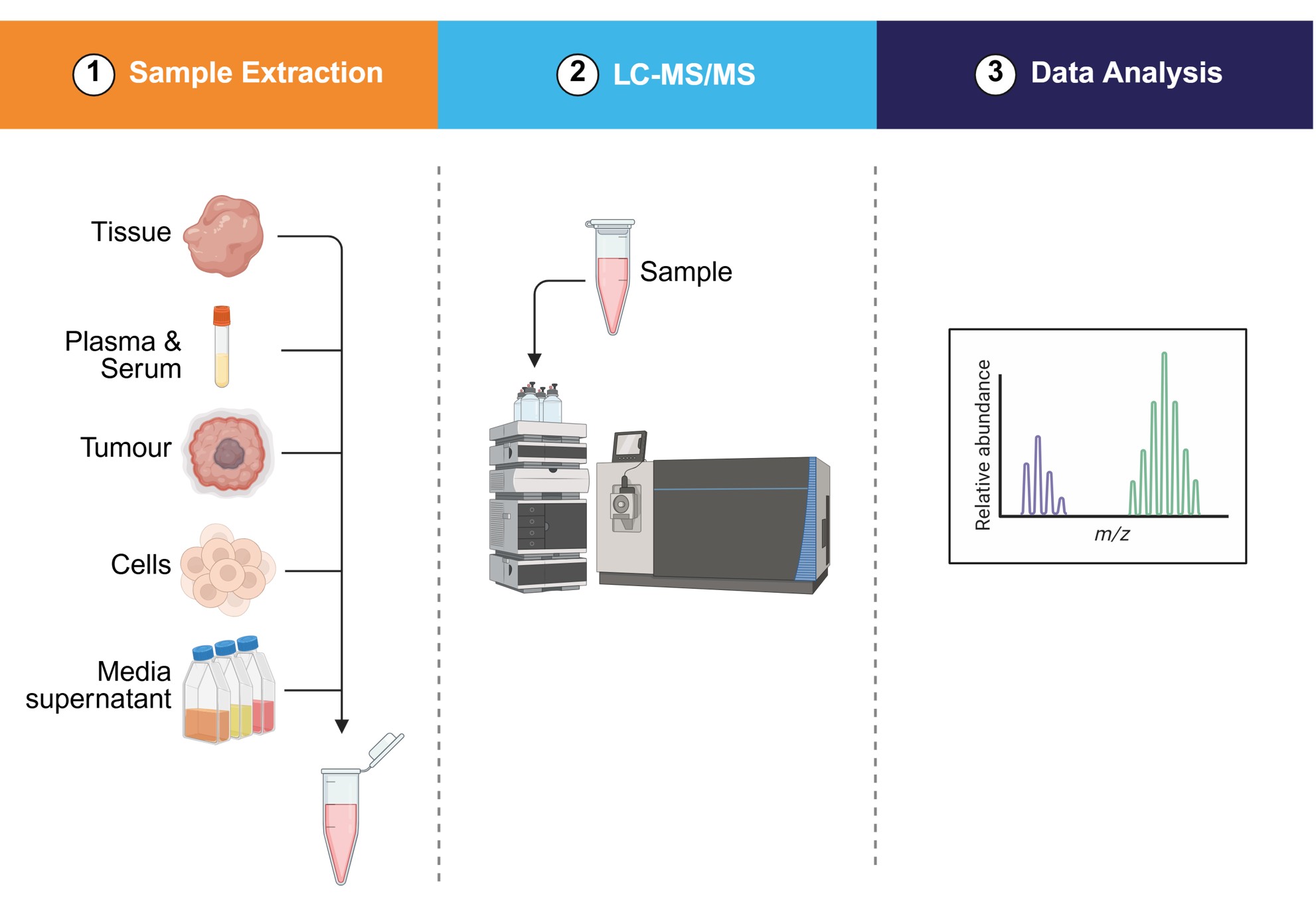
Why choose Concept Life Sciences for biomarker services?
Custom workflows that deliver results fast: Bespoke assay design and optimization tailored to your endpoints.
Versatile expertise across all sample types: From blood and urine to complex tissues and in vitro systems, we work across a range of biological matrices and adapt quickly to evolving project needs.
Technology-driven, outcome-focused: We leverage advanced technologies and scientific insight.
Collaborative partnership: Direct scientist-to-scientist dialogue and transparent data interpretation.
Regulatory confidence: Supporting preclinical and clinical programs worldwide.
Download the Poster: Novel assays to enhance spatial biomarker discovery to learn how our team enhances discovery through advanced imaging and quantification.
Advance your biomarker strategy
From custom biomarker discovery to full clinical trial support, Concept Life Sciences is your trusted partner in drug development, providing scientific depth, operational flexibility and regulatory assurance to accelerate your development journey. Working together to reduce risk, streamline your process and deliver data you can trust.
Contact us today to discuss your biomarker goals and how we can help you design a strategy that reduces risk and drives smarter, faster drug development.
Biomarker FAQs
Q: What are biomarkers in drug development?
A: Biomarkers are measurable biological indicators that reflect disease presence, mechanism of action, or therapeutic response. They enable data-driven decisions at every stage of drug development.
Q: What types of samples can you analyze?
A: We analyze a wide range of biological matrices including blood, plasma, urine, tissue, and cell culture systems — using validated methods tailored to your program.
Q: Do you provide regulatory-compliant bioanalysis?
A: Yes. Our bioanalytical laboratories operate under GLP and GCP conditions, providing validated biomarker quantification suitable for regulatory submission.
Q: How can biomarker services accelerate drug development?
A: Early integration of biomarker strategy reduces uncertainty, improves patient selection, and increases clinical success rates by delivering faster, more confident decision-making.
Contact our biomarker specialists today to enhance your drug development pipeline.
